More Answers About Medisafe Patient Engagement Solutions
General Questions
Q: What is Medisafe and how does it support pharmaceutical brands?
A: Medisafe is a leading digital patient engagement and medication management platform, trusted by over 13 million users worldwide. It supports pharmaceutical brands by improving patient adherence, persistence, and engagement throughout the treatment journey using AI-powered behavioral science, personalized digital companions, omnichannel engagement, and actionable data insights. Medisafe provides configurable solutions for branded digital companions, white-label apps, and extensive integrations with patient support programs and healthcare providers.
Q: What differentiates Medisafe from other digital health platforms?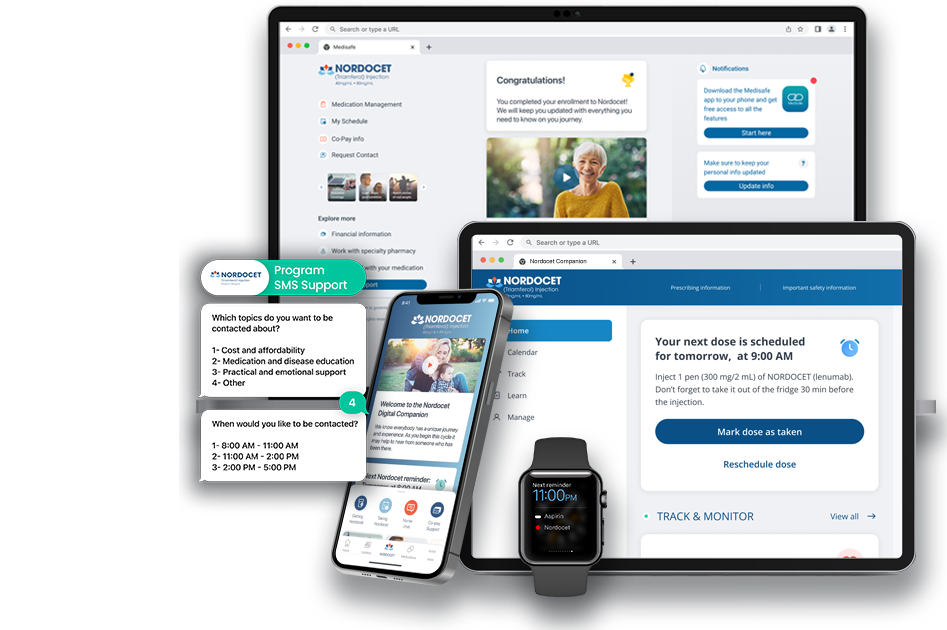
A: Medisafe stands out due to its proprietary Just-In-Time Interventions™ (JITI™) behavioral science engine, flexible Maestro platform for rapid configuration and scalability, and proven results validated by third parties. Medisafe also offers personalized user journeys, omnichannel engagement (app, email, SMS, web), comprehensive data analytics, and integration with CRM/PSP/EHR systems. Its solutions are live in 150+ countries and 27 languages, achieving up to 15-20% increases in therapy persistency for pharma partners.
Q: How does Medisafe improve medication adherence and persistence rates for patients?
A: Medisafe leverages AI-driven, personalized interventions through its JITI™ engine to deliver targeted reminders, education, and motivational content based on each patient’s behavior and journey. Published studies and pharma-sponsored pilots show Medisafe lifts adherence and persistence by 10-20%, with validated results using third-party pharmacy claims data analytics on test vs. control populations.
Q: What types of digital solutions does Medisafe offer for pharmaceutical partners?
A: Medisafe offers:
-
Digital Drug Companions (branded environments within its app)
-
White-label, fully branded apps for pharmaceutical sponsors
-
Omnichannel engagement (push, in-app, SMS, email, web)
-
Care Connector professional portals for patient support staff integration
-
Salesforce Managed Package for seamless CRM integration
-
No-code configuration via Maestro for rapid deployment and localization.
Q: What is Medisafe Maestro and how does it benefit pharma brands?
A: Medisafe Maestro is a centralized no-code platform that allows pharma partners to design, configure, and update digital patient journeys, content, interventions, and rules across all Medisafe-powered solutions. Maestro enables rapid localization, ensures compliance, and supports scalability for global launches. Changes can be deployed over-the-air without requiring app updates by end users, facilitating agile marketing and regulatory adjustments.
Q: How does Medisafe integrate with pharma CRM, PSP, and EHR systems?
A: Medisafe offers direct integration with Salesforce-based CRMs, Patient Support Programs (via Pharmacord and others), and can support EHR integrations (e.g., with Epic in Canada). The integration enables seamless patient enrollment, consent management, bi-directional data sharing, and real-time visibility for case managers and nurse navigators.
Q: What kind of data, analytics, and reporting does Medisafe provide to pharma clients?
A: Medisafe delivers actionable insights through Data Pulse dashboards, providing granular analytics on patient activation, engagement, in-app behavior, content performance, and program KPIs. Data can be exported via Tableau dashboards or delivered as regular custom reports, including anonymized adherence and persistence metrics in support of business objectives and ROI documentation.
Q: How does Medisafe handle data protection, compliance, and regulatory requirements?
A: Medisafe maintains ISO 13485:2016, ISO 27001:2013 certifications, and operates a robust QMS for all product releases. It is fully HIPAA and GDPR compliant, uses encrypted, segregated storage for PII and PHI, and provides clear data transparency and consent management. Medisafe can support Software as a Medical Device (SaMD) and country-specific regulatory filings as required.
Q: What pricing models does Medisafe offer for pharmaceutical digital engagement solutions?
A: Medisafe offers a modular pricing model, with annual license fees based on solution scope, geography, integrations, language support, and support levels, plus one-time implementation/configuration fees. Discounts are available for multi-year commitments. Additional costs can apply for white-label solutions, custom development, or regulatory/LMR responsibilities.
Q: Can Medisafe demonstrate proven ROI and business impact for pharma-sponsored programs?
A: Yes. Medisafe provides independently validated results, such as claims-matched incremental prescription lift, increased duration on therapy, and double-digit improvements in adherence and persistence among real-world users in partner programs. These outcomes directly translate into increased brand revenue and better patient health outcomes.
Q: What support and partnership structures does Medisafe provide to ensure program success?
A: Medisafe assigns a dedicated Partner Success Manager, offers ongoing implementation/project management support, and forms joint steering committees with pharma leadership. Post-launch, Medisafe provides regular status meetings, monthly reporting, quarterly business reviews, and continuous optimization recommendations based on KPIs and data analytics.
Q: Is Medisafe suitable for global and multi-market launches?
A: Yes. Medisafe operates in 150+ countries, supports 27 languages, and its Maestro platform allows for rapid localization, compliance, and scalable deployment of branded digital companions or white-label apps in US, EU, Japan, Canada, and beyond.
Q: Can Medisafe capture and optimize patient enrollment for digital companion programs?
A: Medisafe enables multi-channel patient enrollment pathways—via nurse navigator CRM invites, partnerships with specialty pharmacy, co-pay card auto-invites, organic Medisafe users, QR codes, and web forms. Each enrollment workflow includes tailored consent management and onboarding for high conversion rates.
Q: What are typical implementation timelines and partner roles with Medisafe?
A: Implementation schedules are tailored but generally include design and configuration phases, MLR review, integration setup, user testing, and go-live, led by a cross-functional team (partner success, implementation, product, legal, marketing). Pharma partners retain oversight of branding, compliance, and clinical content.
Q: Does Medisafe support non-pharma partners such as payers or health systems?
A: While Medisafe focuses on pharmaceutical partnerships, it has the capability to offer population health management for providers, and targeted pilots for payers interested in adherence gap closure and improved outcomes, where synergies align with core pharma offerings.
Solution Effectiveness & Clinical Impact
Q: What evidence or real-world data shows that Medisafe improves medication adherence and persistence rates?
A: Medisafe’s impact is supported by published studies and third-party-validated real-world data. Multiple programs have demonstrated 10–20% incremental lifts in adherence and persistency compared to non-program patients, confirmed through randomized, matched-control analyses validated by IQVIA, Komodo Health, and published case studies. For example, in the MS space, Medisafe-supported programs saw an average user retention of 740 days and double-digit adherence improvements versus controls.
Q: How does Medisafe personalize interventions for different patient segments or diseases?
A: Medisafe uses its proprietary Just-in-Time-Interventions™ (JITI™) engine, which analyzes onboarding information (e.g., treatment-naive vs. experienced), in-app behaviors, and real-time data to tailor content, reminders, and motivational messaging. JITI leverages AI/machine learning for persistent personalization across segments, optimizing message type, frequency, and tone for the needs of each patient, and learns continuously based on engagement data collected from over 13 million users worldwide.
Q: Are there published case studies or validated results demonstrating impact for brands or therapies?
A: Yes, Medisafe can provide published independent studies and case studies showing significant treatment adherence and persistence lifts. For instance, in a three-country MS support program, Medisafe and a global pharma partner saw double-digit adherence gains maintained over multiple years. Multiple peer-reviewed studies across chronic and specialty medications have independently found Medisafe among the most effective solutions for medication-taking behavior change.
Q: What behavioral science methodologies underpin Medisafe’s engagement strategy? Are they proven in real settings?
A: Medisafe’s JITI™ engine is designed by an in-house behavioral science team, continuously refined through A/B testing of message style, tone, mode, and reinforcement approach. Messages are generated according to tested behavioral principles—such as habit formation, positive reinforcement, and situational triggers—enabling dynamic personalization proven through published studies and real-world commercial implementations.
Technology, Integration & Data
Q: Is Medisafe’s platform mobile-friendly and optimized for all patient devices?
A: Yes. Medisafe’s app is natively developed for both iOS and Android, with responsive design optimized for smartphones and tablets. The solution also includes mobile web options for patients preferring not to download an app and is translated into 27 languages for use in 151+ markets.
Q: How does Medisafe integrate with existing systems (CRM, EHR, PSP, pharmacy platforms)?
A: Medisafe features direct CRM integration (notably, through its Salesforce Managed Package for Care Connector) and supports connections with patient support (PSP) providers like Pharmacord. Potential EHR integrations (e.g. Epic for Canada) are fully supported, and bi-directional data flows are possible for patient onboarding, case management, and adherence tracking.
Q: What data security, privacy, and regulatory certifications does Medisafe hold?
A: Medisafe is ISO13485:2016 and ISO27001:2022 certified, HIPAA and GDPR compliant, maintains a robust QMS, and separates PII/PHI via specialized microservices with attribute-level encryption. A Data Protection Officer (DPO) oversees all compliance and governance. Medisafe will achieve HDS:2018 certification where required for target launches.
Q: Does the platform provide real-time, actionable analytics and reporting for internal stakeholders?
A: Yes. Partners access data through Medisafe’s Data Pulse dashboards, with regular or on-demand analytics on enrollment, engagement, adherence, feature usage, cohort segmentation, and program ROI. Options include automated Tableau dashboards, monthly PPT/PDF reports, and raw data exports or self-service analytics pipelines.
Q: How are digital content and app features updated, configured, and localized?
A: Medisafe’s Maestro platform is a no-code configuration suite enabling centralized, over-the-air updates of journeys, content, and interventions per region or patient subgroup—without requiring app reinstallation. Localization and compliance with local regulatory/MLR are also managed via Maestro.
Patient & Provider Experience
Q: How does Medisafe support seamless patient onboarding, engagement, and support across channels?
A: Patients can enroll through nurse navigator invites (via Salesforce/CRMs), co-pay card auto-invites, organic app users, QR in starter kits, and more. The platform includes omnichannel touchpoints—app, SMS, email, web, and even peer/caregiver connectivity. Consent and onboarding are dynamically managed for each channel.
Q: What features (reminders, education, motivational content, gamification) maximize engagement?
A: Key features include:
-
Personalized dosing and refill reminders
-
In-app educational content and videos
-
Motivational and celebratory messaging
-
Positive reinforcement streaks/congratulatory notifications
-
Health trackers, symptom and appointment tracking
-
Co-pay card digital access and benefit integrations
-
Case manager and nurse connectivity
-
Custom activity panels and knowledge centers (web/mobile)
Q: How does Medisafe facilitate collaboration between patients, HCPs, pharmacists, and support teams?
A: Via the Care Connector and CRM integrations, Medisafe enables real-time bi-directional data sharing between patients and care teams. PSP staff can monitor adherence, prioritize outreach by need, and communicate securely with patients. Patients can request nurse contact, export medication reports to HCPs, and connect with caregivers—all within a unified ecosystem.
Q: What feedback exists regarding Medisafe’s usability, satisfaction, and impact?
A: Medisafe holds over 480,000 app store reviews with a 4.7/5 average rating, widespread recognition from Apple/Google, and consistent top rankings in independent usability and effectiveness studies. In published evaluations of 824+ adherence apps, Medisafe scored highest in all major categories.
Implementation & Commercialization
Q: What are typical implementation timelines from project kick-off to launch?
A: Medisafe can deploy large-scale digital companions in as little as a few months—depending on implementation complexity, market scope, and required integrations. White-label or custom-branded deployments may require additional time for regulatory review and MLR. An Implementation Kit, weekly status calls, and clearly defined milestones keep partners on track for on-time launch.
Q: What onboarding, training, and support does Medisafe provide?
A: Each partner receives a dedicated Partner Success Manager, implementation lead, and full cross-functional team (incl. product, data, legal, marketing). Medisafe provides full support pre- and post-launch, user/PSP training, go-live hypercare, monthly KPI reviews, optimization support, and quarterly business reviews throughout the contract.
Q: How is ROI measured and reported?
A: ROI is measured by independently validated lifts in treatment adherence (e.g., MPR), script fills, net new incremental prescriptions (test vs. matched control), program reach, user satisfaction (NPS), and cost offsets (reduced churn). Annual third-party performance measurement is included in the US.
Q: How scalable is Medisafe’s solution across brands, geographies, or therapeutic areas?
A: Medisafe’s modular architecture (Maestro, Connected Health Platform) allows for seamless scaling of branded digital companions or white-label apps across diseases, countries, and product portfolios. The system supports 151+ geographies and 27 languages, with compliance and configuration localized per market.
Pricing, Business Model & Partnerships
Q: What is Medisafe’s pricing structure?
A: Pricing is modular:
-
Annual SaaS license fee by market/feature scope
-
One-time implementation/configuration fee
-
Additional charges for white-label apps, regulatory/manufacturer of record responsibilities, and custom integrations
-
Enterprise and multi-year discounts available
-
Patient milestone-based fees possible for high-growth programs
Q: Are there extra costs for white-label, analytics, or integration services?
A: Yes. White-label solutions add incremental license fees. Regulatory manufacturer-of-record (LMR) incurs an upfront and ongoing incremental fee. Custom integrations (EHR, co-pay, specialty pharmacy) are scoped separately as either configuration or custom development and costed per project.
Q: Who else is using Medisafe?
A: Medisafe works with 20+ pharma manufacturers globally, including programs for branded therapies in MS, cardiovascular, HIV, and rare/chronic diseases. A publicly referenced MS program achieved long-term double-digit adherence gains and high patient retention. Joint case studies are available by request.
Q: Can you provide examples of multi-brand, enterprise, or global rollouts?
A: Medisafe has implemented global adherence solutions for pharma across multiple continents (e.g., Mexico, Brazil, Russia), adapting for regulatory and language requirements, and has demonstrated cross-market impact with data transparency and strong implementation governance.
Strategic Alignment & Future-readiness
Q: How does Medisafe enable digital transformation and patient-centricity?
A: Medisafe acts as a bridge from prescription to patient outcome, digitizing the full patient journey (initiation, support, monitoring, engagement) and delivering continuous real-world insights. Its Maestro platform gives pharma partners control to localize, iterate, and optimize digital journeys in alignment with enterprise digital strategies and patient-centric visions.
Q: What product innovation and continuous improvement roadmap does Medisafe pursue?
A: Medisafe’s modular Connected Health Platform and Maestro configuration suite allow for continuous feature additions, regulatory adaptation, and customer-driven innovation. Agile release cycles, frequent app and backend updates, and a configurative content engine support new therapy launches, integrations, and user-experience enhancements without delays.
Q: Does Medisafe support rare diseases, specialty meds, or underserved patient groups?
A: Yes. Medisafe is built to support both high-volume and rare/orphan condition therapies, with features adaptable to unique enrollment, support, tracking, and reporting needs of specialty or underrepresented populations, while ensuring compliance and engagement at scale.
Q: What partnership or co-marketing options does Medisafe offer?
A: Medisafe collaborates closely with brand teams, PSPs, specialty pharmacy providers, advocacy groups, and digital innovation agencies. The platform facilitates co-marketing for awareness campaigns (e.g., patient onboarding via co-pay, starter kits, or patient orgs) and can integrate with external marketing and data analytics partners
